
Anne McNeil, University of Michigan and Madeline Clough, University of Michigan
It seems like every day a new study finds tiny plastic particles called microplastics where they should not be: in our bodies and our food, water and air.
Yet finding and identifying microplastics is extremely challenging, especially given their small size. One microplastic can range from as large as a ladybug to as small as an eighth of a red blood cell.
In addition, it can be hard for researchers to avoid unintentionally contaminating their samples, because these plastics are practically everywhere. As a result, much of this research may be overestimating the number of microplastics.
In a new study published in March 2026, our team found that, even when following established protocols, using certain methods to measure environmental microplastics can potentially contaminate the results.
The study
We are chemists at the University of Michigan working in a collaborative team. We set out to understand how many microplastics Michiganders were inhaling when outside, and whether that depended on where they lived.
When preparing our samples, we followed all the standard protocols while conducting our research – we avoided plastic use in the lab, wore nonplastic clothing and even used a specialized chamber to reduce potential contamination from the laboratory air.
Despite these precautions, we found plastic counts in the air that were over 1,000 times greater than previous reports. We knew these numbers didn’t seem right, so what happened?
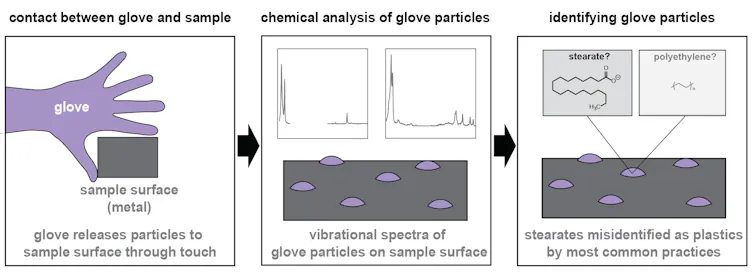
The culprit: Lab gloves
After a long path to pinpointing the contamination source, we found that laboratory gloves, which the scientific community recommends using as a best practice, can transfer particles to the surface of our samples – in this case, small metal sheets used to collect material depositing from the air. Moreover, the particles led to an overestimation of microplastic abundance in our study.
Here’s how: The particles, which we identified as stearate salts, are used to help the gloves cleanly release from their mold during the manufacturing process. When gloves are used to handle laboratory equipment, the particles are transferred to anything they touch. Stearate salts are similar to soap molecules – if you eat a lot of them, they’re probably not good for you, but they’re not harmful in the environment in the same way that microplastics are.
While not microplastics themselves, stearate salts are structurally similar to polyethylene, the type of plastic most often found in the environment. This structural similarity makes it difficult to distinguish them using the most common tools scientists use to determine whether a particle is plastic.
Researchers use vibrational spectroscopy to identify microplastics, which entails measuring how the particle interacts with light to produce what scientists call a chemical fingerprint.
Because polyethylene and stearate salts have very similar structures, they also interact with light in a similar way.
As a result, at least some of the time, the particles from gloves are incorrectly identified as microplastics. As more researchers rely on automated methods to speed up their analyses, glove residue may be increasingly mistaken for microplastics, leading to higher reports of microplastics in the environment than in reality.
How widespread is this contamination?
To investigate how prevalent this contamination might be, we looked at different glove types. We mimicked the touch between seven types of gloves while handling laboratory equipment and counted the number of microplastics we would incorrectly attribute to the environment if we followed the most common approaches.
We found that gloves can contribute over 7,000 particles per square millimeter that are misidentified as microplastics. This finding means that researchers could be unknowingly overestimating microplastic abundance in the environment when handling their samples with gloves.
Even more concerning, we found that the particles were largely less than 5 um in size. Microplastics in this size range have larger impacts on human and ecosystem health because they can more easily enter cells. By inflating microplastic counts in this size range, using laboratory gloves may jeopardize the studies that inform future policies and regulations.
Moving forward
To avoid contamination, we suggest scientists avoid glove use while conducting microplastic research. If that is not possible – for example, with biological samples where the researchers must wear gloves to protect themselves – we recommend a glove made without stearates, such as those designed for electronics manufacturing. To recover older, potentially contaminated datasets, we have developed methods to help differentiate the chemical fingerprints.
Science is an iterative process. New areas of research, including environmental microplastics, introduce new challenges to the scientific community. In addressing these new challenges, we will encounter setbacks, such as unforeseen contamination.
While we had to discard our initial dataset, we expect the lessons we learned about glove contamination to reach other scientists. In addition, we plan to continue our research on Michigan’s atmospheric microplastic contamination – but this time without gloves.
It’s important to note that even if the microplastic abundance in the environment is lower than researchers originally thought, any amount of microplastics can be troublesome, given their negative effects on human health and ecosystems.
Anne McNeil, Professor of Chemistry and Macromolecular Science and Engineering, University of Michigan and Madeline Clough, Ph.D. Candidate in Chemistry, University of Michigan
This article is republished from The Conversation under a Creative Commons license. Read the original article.




















