
Pieter Visscher, University of Connecticut; Brendan Paul Burns, UNSW, and Kimberley L. Gallagher, Quinnipiac University
Billions of years ago, life on Earth was mostly just large slimy mats of microbes living in shallow water. Sometimes, these microbial communities made carbonate minerals that over many years cemented together to become layered limestone rocks called stromatolites. They are the oldest evidence of life on Earth. But the fossils don’t tell researchers the details of how they formed.
Today, most life is supported by oxygen. But these microbial mats existed for a billion years before oxygen was present in the atmosphere. So what did life use instead?
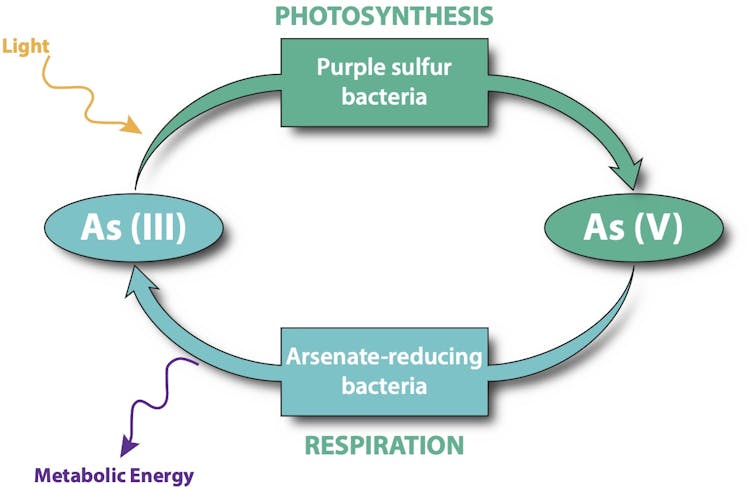
Our team of geologists, physicists and biologists had found hints in fossilized stromatolites that arsenic was the chemical of choice for ancient photosynthesis and respiration. But modern-day versions of these microbial communities still live on Earth today. Perhaps one of these used arsenic and could offer proof for our theory?
So we joined a surveying expedition of Chilean and Argentinian scientists to look for living stromatolites in the extreme conditions of the High Andes. In a small stream deep in the Atacama Desert, we found a big surprise. The bottom of the channel was bright purple and made of stromatolite-building microbial mats that thrive in the complete absence of oxygen. Just as the clues we’d found in ancient fossils suggested, these mats use two different forms of arsenic to perform photosynthesis and respiration. Our discovery offers the strongest evidence yet for how the oldest life on Earth survived in a pre-oxygen world.
Christophe Dupraz, Anthony Bouton, Pieter Visscher, CC BY-ND
Turning sunlight into energy
For the last 2.4 billion years, photosynthetic organisms like plants and blue-green cyanobacteria have used sunlight, water and carbon dioxide to make oxygen and organic matter. In doing this, they turn energy from the Sun into energy to be used by life. Other organisms breathe in oxygen as they digest organic carbon, gaining energy for their respiration in the process.
Microbes in the ancient world also captured energy from sunlight, but their primitive machinery could not make oxygen from water or use oxygen for respiration. They needed another chemical to do this.
From a biochemical perspective, there are only a few possible candidates: iron, sulfur, hydrogen or arsenic. A lack of evidence in the fossil record and minuscule amounts of some of these chemicals in the primordial soup suggests neither iron, sulfur nor hydrogen would be likely candidates for the earliest form of photosynthesis. That leaves arsenic.
In 2014, our team found the first clue that stromatolites were produced by arsenic-assisted photosynthesis and respiration. We collected pieces of 2.72-billion-year-old stromatolites from the pre-oxygen world by drilling into an ancient reefs in the Outback of Australia. We then took these samples to France and cut them into thin slivers. By measuring the X-rays that came off these samples when we bombarded them with photons, we made a map of the chemical elements in the sample. If two kinds of arsenic are present in the map, then it is a sign that life was using arsenic for photosynthesis and respiration. In these relics of ancient life we found lots of both forms of arsenic, but not iron or sulfur.
This was tantalizing, but we wanted more proof: a modern analog to help prove our arsenic theory. No researchers had ever found a microbial mat community living in a place completely free of oxygen, but if we found one, it could help explain how the first stromatolites formed when our planet’s oceans and atmosphere were lacking oxygen.

D’Angelo Duran, CC BY-ND
Modern microbes, ancient analogs
The Atacama Desert in Chile is the driest place on Earth, flanked by volcanoes and exposed to extremely high UV radiation. It’s not too different from how the Earth looked 3 billion years ago and not exactly supportive of life as we know it. Here – with the help of a team that spanned four continents and seven countries – we found what we were looking for.
Or destination was Laguna La Brava, a very salty shallow lake deep into the harsh desert. A shallow stream, fed by a volcanic groundwater spring, led into the lake. The streambed was a unique, deep purple color. The color came from a microbial mat, thriving quite happily in waters that contained unusually high amounts of arsenic, sulfur and lithium, but missing one important element – oxygen.
Could these slimy purple blobs offer answers to an ancient question?

Pieter Visscher, CC BY-ND
We cut a piece of the mat and looked for evidence of minerals. A drop of acid made the minerals fizz – carbonates! – this microbe community was forming stromatolites. So our team went to work, camping out at the site for days at a time.
We measured the chemistry of the water and the mat with our field equipment during day and night, summer and winter. Not once did we find oxygen, and back in the laboratory we confirmed that sulfur and arsenic were abundant. Looking through the microscope, we saw purple photosynthetic bacteria, but oxygen-producing cyanobacteria were eerily absent. We had also collected DNA samples from the mat and found genes for arsenic metabolism.
In the lab, we mixed up microbes from the mat, added arsenic and exposed the mix to sunlight. Photosynthesis was happening. The microbes used both arsenic and sulfur, but preferred the arsenic. When we added a minuscule amount of organic matter, a different arsenic compound was used for respiration and preferred over sulfur.
[You’re too busy to read everything. We get it. That’s why we’ve got a weekly newsletter. Sign up for good Sunday reading. ]
All that was left was to show that the two types of arsenic could be detected in the modern stromatolites. We went back to France, and using an X-ray emission technique made chemical maps from the Chilean samples. Every experiment we performed supported the presence of a vigorous arsenic cycle in the absence of oxygen in this unique modern stromatolite. This validates, beyond doubt, the idea that the fossil Australian samples that we studied in 2014 held evidence of an active arsenic cycle in deep time on our young planet.

Pieter Visscher, CC BY-ND
Answers on Earth, leads for Mars
The harsh conditions of the Atacama are so similar to Martian and early Earth environments that NASA scientists and astrobiologists turn to the Atacama to answer questions about how life began on our planet, and how it might start elsewhere. The arsenic-cycling mats we discovered at Laguna La Brava offer strong clues to some of the most fundamental questions about life.
On board the Mars 2020 Perseverance rover that is currently hurtling through space is an instrument that can observe elements using the exact same process we used to make our element maps. Perhaps it will discover that arsenic is abundant in layered rocks on Mars, suggesting that life on Mars also used arsenic. For over a billion years, it did so on Earth. Under the harshest conditions life finds a way, and it is that way we are trying to understand.![]()
Pieter Visscher, Professor of Marine Sciences, University of Connecticut; Brendan Paul Burns, Senior Lecturer, UNSW, and Kimberley L. Gallagher, Adjunct Professor of Chemistry, Quinnipiac University
This article is republished from The Conversation under a Creative Commons license. Read the original article.




















